Tag: bioequivalence
Therapeutic Equivalence Codes: How the FDA Rates Generic Drug Substitutability
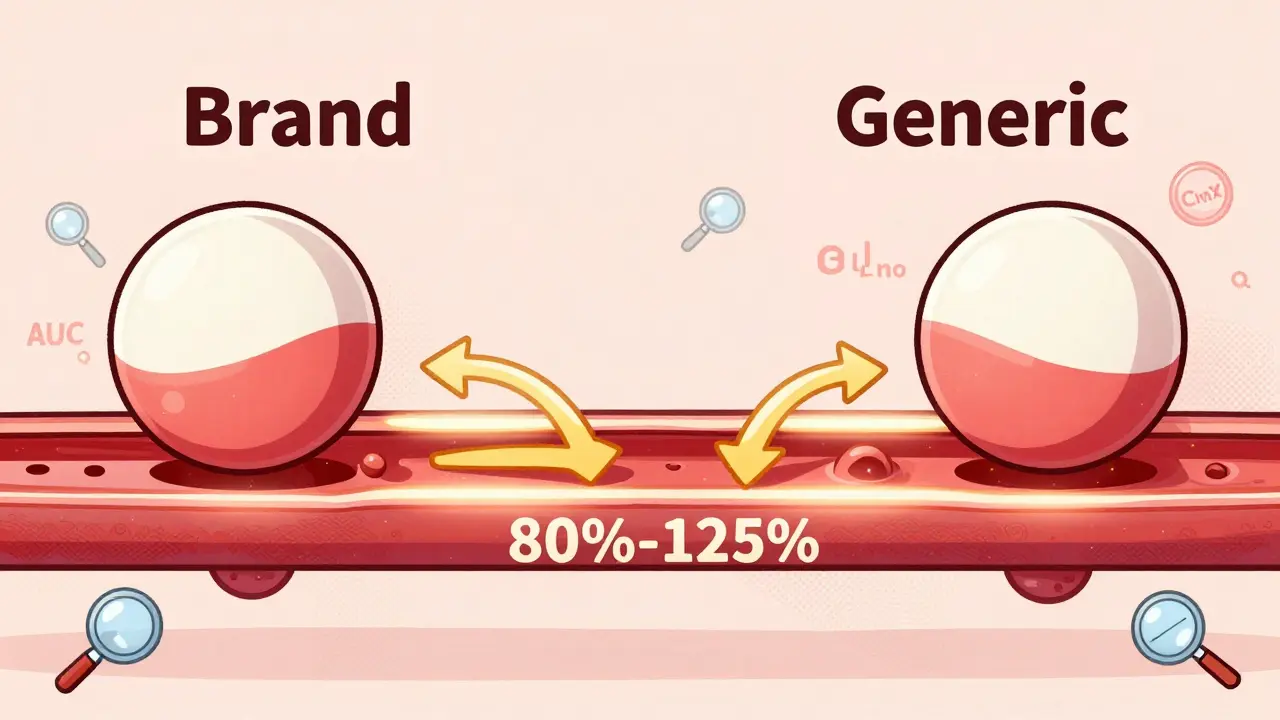
The FDA's therapeutic equivalence codes tell pharmacists which generic drugs can be safely swapped for brand-name versions. Learn how the system works, why some drugs get "B" ratings, and how it saves billions in healthcare costs.
View MoreBioequivalence and Patient Safety: Why Testing Ensures Safe Generic Medications
Bioequivalence testing ensures generic drugs work the same as brand-name versions, protecting patient safety while cutting costs. Learn how science, regulation, and real-world data confirm their reliability.
View MoreHow to Compare Dissolution Profiles and What They Mean for Generic and Brand Drugs
Learn how dissolution profile comparison ensures generic drugs work like brand-name versions, using f2 similarity factors, regulatory standards, and real-world testing methods to prove bioequivalence without human trials.
View More