The reality is that quality defects in generics is a recurring issue where pharmaceutical products deviate from their intended formulation, resulting in compromised safety, efficacy, or stability. These failures are often the hidden driver behind drug shortages. In fact, data from the FDA Drug Shortages Task Force shows that quality problems caused 62% of drug shortages between 2013 and 2017. When a manufacturing plant fails an inspection, the supply chain breaks, and patients are left without critical medication.
Why Generics Face More Quality Hurdles
You might wonder why generic versions of drugs seem more prone to these issues than the original brand-name versions. It usually comes down to the bottom line. Generic manufacturers operate in a high-pressure environment where price competition is fierce. To keep costs low, some companies cut corners on quality investment. On average, branded companies spend about 15-18% of their production costs on quality assurance, while generic firms often only allocate 8-10%.
Beyond the budget, there's the issue of aging infrastructure. Many generic drugs are produced in facilities using outdated machinery that simply wasn't designed for today's precision standards. Additionally, many of these plants use shared production lines to make multiple different drugs. While efficient, this increases the risk of cross-contamination, where trace amounts of one medicine end up in another. This systemic pressure is why generic facilities have failed FDA inspections at nearly double the rate of branded plants-57% compared to 28%.
Common Physical Defects in Oral Solids
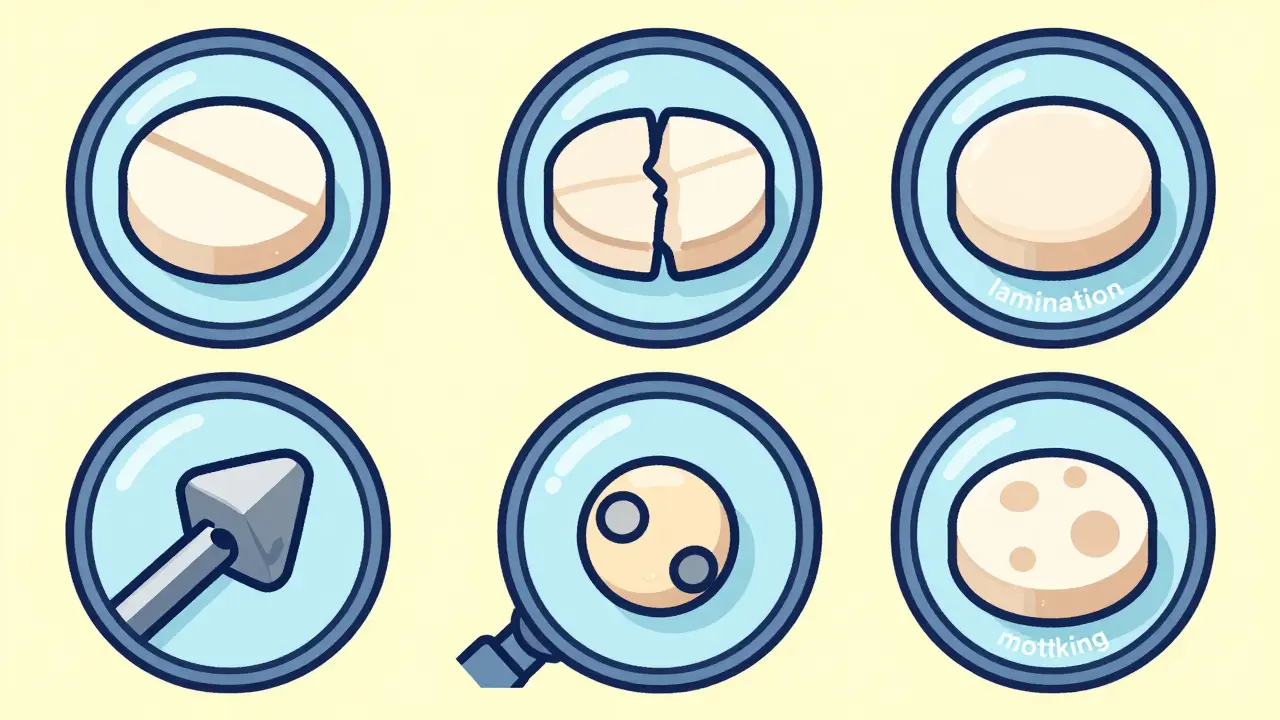
Most people interact with generics through tablets or capsules. When these fail, the defects usually fall into a few specific categories. These aren't just "mistakes"; they are the result of specific physics and chemistry failing during the compression process.
- Capping: This is when the top or bottom crown of the tablet separates horizontally. It often happens when the compression force is too high (over 15 kN) or the mixture is too dry (below 2% moisture).
- Lamination: Similar to capping, but the tablet separates into multiple layers. This usually happens when the machine's turret spins too fast-above 40 rotations per minute-without enough pre-compression to lock the powder in place.
- Sticking: This occurs when the tablet material clings to the punch heads of the machinery. It's common with ingredients that have a low melting point (under 120°C) or when moisture levels climb above 4%.
- Mottling: This is the uneven distribution of color across the tablet surface, which can make a drug look "spotted" and erode patient confidence in the product's consistency.
| Metric | Generic Manufacturers | Branded Manufacturers |
|---|---|---|
| QA Budget (% of production cost) | 8-10% | 15-18% |
| FDA Inspection Failure Rate | 57% | 28% |
| Recall Rate (per facility) | 2.3x higher | Baseline |
| Manufacturing-led Recalls (%) | 63% | 41% |
Critical Failures: Dose Uniformity and Contamination
While a chipped tablet is annoying, "critical defects" are the ones that truly endanger patients. The most dangerous is weight variation. If a tablet is too light or too heavy, the patient isn't getting the correct dose. According to USP <905> is the United States Pharmacopeia standard for Uniformity of Dosage Units , weight variation becomes a failure when granule flow rates drop below 0.5 g/s. In some affected batches, this leads to dose uniformity failures in nearly 13% of the product.
Then there are sterile injectables. These are high-risk because they bypass the body's natural defenses. The primary defect here is particulate contamination-tiny pieces of glass, metal, or plastic getting into the vial. While oral solids have a higher overall defect rate (12.4%), the 8.7% defect rate in injectables is far more concerning because the stakes are higher. A particle in a vein can cause an embolism or severe inflammatory response.
The Regulatory Response and Modern Fixes
Regulators aren't just standing by. The FDA and EMA are pushing for Current Good Manufacturing Practice (also known as CGMP) is a system for ensuring that products are consistently produced and controlled according to quality standards . When these standards are ignored, it leads to the 24% of recall events tied to CGMP deficiencies.
The industry is moving toward "Quality by Design" (QbD). Instead of just testing the final pill to see if it's okay, manufacturers are now required to define a "design space." This means they establish exact operating ranges (usually ±15%) for every part of the process. If a machine drifts outside that range, it's flagged immediately.
We're also seeing a shift toward automated visual inspection. Humans are surprisingly bad at spotting tiny defects; error rates can be as high as 30%. New AI-powered systems can detect flaws as small as 0.1 mm at a speed of 600 tablets per minute, cutting the error rate to under 2%. Sandoz and Dr. Reddy's have already piloted AI systems that boast 92% detection accuracy, far surpassing traditional manual checks.

Real-World Impact: What Pharmacists and Patients See
This isn't just a theoretical problem for engineers. Pharmacists are on the front lines. A survey of over 1,200 pharmacists found that 68% encountered generic quality issues in a single year. They report things like Metformin ER tablets crumbling during the dispensing process or patients claiming that a generic version of Levothyroxine feels "different" despite coming from the same manufacturer.
These experiences lead to a loss of trust. In 2023, nearly 10% of generic substitution requests in hospitals resulted in a permanent switch back to the brand name because of quality concerns. When a patient sees a chipped or discolored pill, they stop trusting the medicine, which can lead to them skipping doses or stopping treatment entirely.
Are generic drugs less effective because of these defects?
Not necessarily. Most generics are bioequivalent to the brand. However, manufacturing inconsistencies-such as weight variation or poor dissolution-can lead to "therapeutic inequivalence" in a small percentage of batches, meaning the drug may not work as consistently as the brand name.
How can I tell if my generic medication has a quality defect?
Look for physical abnormalities: tablets that are splitting (capping), peeling (lamination), or have uneven coloring (mottling). If pills are crumbling or have an unusual smell or color change, contact your pharmacist immediately.
Why are there so many recalls for generics compared to brands?
Generics are often produced in higher volumes with tighter profit margins. This leads to lower investment in quality control and the use of older equipment, which increases the likelihood of manufacturing errors that trigger recalls.
What is the "design space" in drug manufacturing?
The design space is a set of pre-determined limits for manufacturing variables (like temperature, pressure, and moisture). As long as the process stays within these limits, the final product is guaranteed to meet quality standards.
Does the FDA monitor these defects in real-time?
The FDA doesn't monitor every machine in real-time, but they conduct periodic inspections and review batch records. They also rely on the MedWatch system where patients and doctors report adverse events and visible defects.
Next Steps for Patients and Providers
If you're a patient and you notice your medication looks odd, don't just toss it. Note the lot number on the bottle and report it to your pharmacist. This data feeds into systems like MedWatch, which helps regulators identify failing plants before a massive shortage occurs.
For healthcare providers, focusing on "complex generics"-like inhalers or modified-release tablets-is key. These have much higher defect rates (18.2% and 14.7% respectively) than simple immediate-release tablets. Being extra vigilant with these specific categories can help catch quality issues before they impact patient health.




Mary Johnson
13 April, 2026 . 03:53 AM
This is just the tip of the iceberg!! They want us to believe it is just "outdated machinery" but it is obviously a coordinated effort to weaken the population with sub-par meds while they rake in billions!!! Who is actually auditing these plants anyway because the FDA is probably in their pockets!!! Wake up people!
john chiong
13 April, 2026 . 08:47 AM
absolute corporate greed at its most putrid. these suits are playing russian roulette with our health just to squeeze another nickel out of the peasants. absolutely abhorrent behavior from the top brass who sleep on silk sheets while we swallow crumbling chalk
Billy Wood
14 April, 2026 . 10:54 AM
Report every single defect!!! Be a champion for health!!! Let's push for better standards!!!
Tabatha Pugh
16 April, 2026 . 06:33 AM
Actually, the USP <905> standard is far more complex than the post suggests, specifically regarding the distinction between content uniformity and weight variation which are not interchangeable terms in a strict pharmaceutical context, though most people here probably don't know the difference.
Princess Busaco
16 April, 2026 . 07:36 AM
It is truly fascinating how some people just blindly trust the "design space" logic as if a mathematical range can magically erase the inherent incompetence of a facility that has already failed an inspection twice, and quite frankly, the idea that AI can replace human oversight is just a desperate attempt by corporations to further dehumanize the medical process while pretending they care about a 0.1 mm defect when the real issue is the systemic rot of the entire profit-driven pharmaceutical complex which is honestly just a tragedy of modern capitalism.
Rim Linda
17 April, 2026 . 11:48 AM
Omg my last batch of meds literally looked like they had been chewed by a dog!! 😱 So scary!!
Haley Moore
17 April, 2026 . 13:44 PM
Imagine actually being surprised that the cheapest option is the worst quality... like, duh? 🙄 It's giving "I just discovered how capitalism works" vibes. Truly exhausted by this discourse 💅
Catherine Mailum
18 April, 2026 . 20:48 PM
oh wow i am just so shocked that companies making drugs for money might have a few tiny errors... truly a tragedy for the ages i might just faint from the horror of a chipped pill
melissa mac
20 April, 2026 . 06:25 AM
I think it is important to remember that generics still save so many people from bankruptcy, so let's try to find a way to support the regulations without completely demonizing the providers who make these meds accessible to everyone.
Clare Elizabeth
20 April, 2026 . 11:02 AM
exactly let's just focus on the progress with AI and QbD because those sounds like amazing ways to make everything safer for everyone in the long run
Jasmin Stowers
22 April, 2026 . 05:41 AM
agree it is all about balance