Autoimmune hepatitis isn’t something you hear about every day, but if you’re living with it, you know how deeply it affects your life. It’s not caused by alcohol, viruses, or fatty foods-it’s your own immune system turning against your liver. The body attacks liver cells like they’re invaders, causing inflammation that can slowly scar the organ over time. Left untreated, it can lead to cirrhosis, liver failure, or even the need for a transplant. The good news? With the right diagnosis and treatment, most people can stop the damage and even reverse some of it.
How Is Autoimmune Hepatitis Diagnosed?
There’s no single test that confirms autoimmune hepatitis. Instead, doctors piece together clues from blood work, symptoms, and a liver biopsy. If you’ve been feeling unusually tired, have dark urine, jaundice, or joint pain, and your liver enzymes are sky-high, that’s your first red flag. Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels often spike to five to ten times the normal range. But elevated enzymes happen in many liver conditions, so doctors have to rule out hepatitis B or C, alcohol use, or drug reactions first.
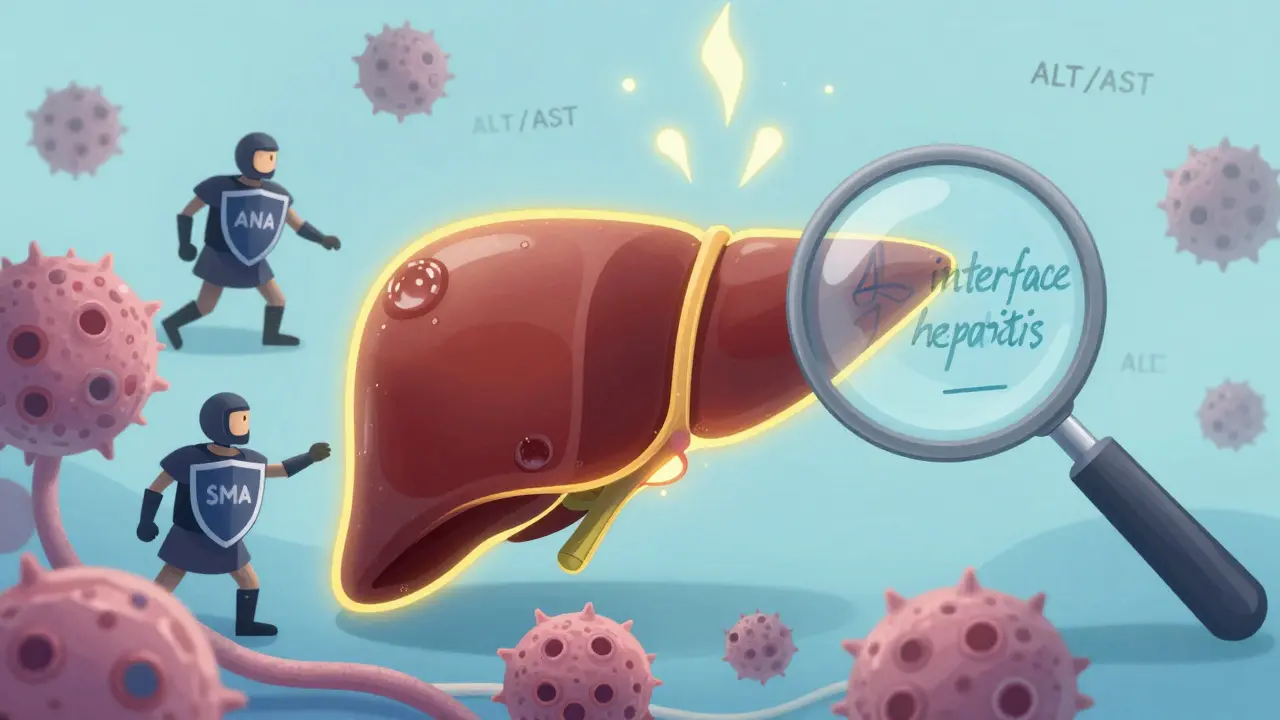
The next step is checking for specific antibodies. Most people with type 1 autoimmune hepatitis-about 80% of cases-have antinuclear antibodies (ANA) or smooth muscle antibodies (SMA) in their blood at titers of 1:40 or higher. Type 2, which is rarer and more common in children, shows liver-kidney microsomal type 1 (LKM1) antibodies. But here’s the key update from the 2025 European Association for the Study of the Liver guidelines: autoantibody types don’t change how you’re treated. Whether you’re ANA-positive or LKM1-positive, your treatment plan stays the same. That’s a big shift from older practices.
The real gold standard for diagnosis is a liver biopsy. This isn’t a routine scan-it’s a small tissue sample taken with a needle, usually guided by ultrasound. The pathologist looks for interface hepatitis: a specific pattern of inflammation where immune cells creep into the liver’s edge, damaging the boundary between the portal areas and liver cells. Experts now say you need to see this pattern in at least 20 portal tracts to confirm the diagnosis. Without this, many cases get misdiagnosed as other liver diseases.
Doctors also use a scoring system called the Revised International Autoimmune Hepatitis Group (IAIHG) criteria. Points are given for things like IgG levels (which are usually 1.5 times higher than normal), the presence of autoantibodies, liver histology, and the absence of viral causes. A score above 15 suggests probable autoimmune hepatitis; above 20 confirms it. This system isn’t perfect, but it helps avoid missing the diagnosis when symptoms are vague.
Why Steroids Are the First Line of Treatment
Since the 1970s, corticosteroids have been the backbone of autoimmune hepatitis treatment. Prednisone or prednisolone works fast-often within two weeks-by calming down the overactive immune system. About 80-90% of patients see their liver enzymes drop significantly in that short time. That rapid response is so consistent that experts say it’s almost diagnostic itself: if your liver enzymes improve quickly on steroids, it strongly supports the diagnosis.
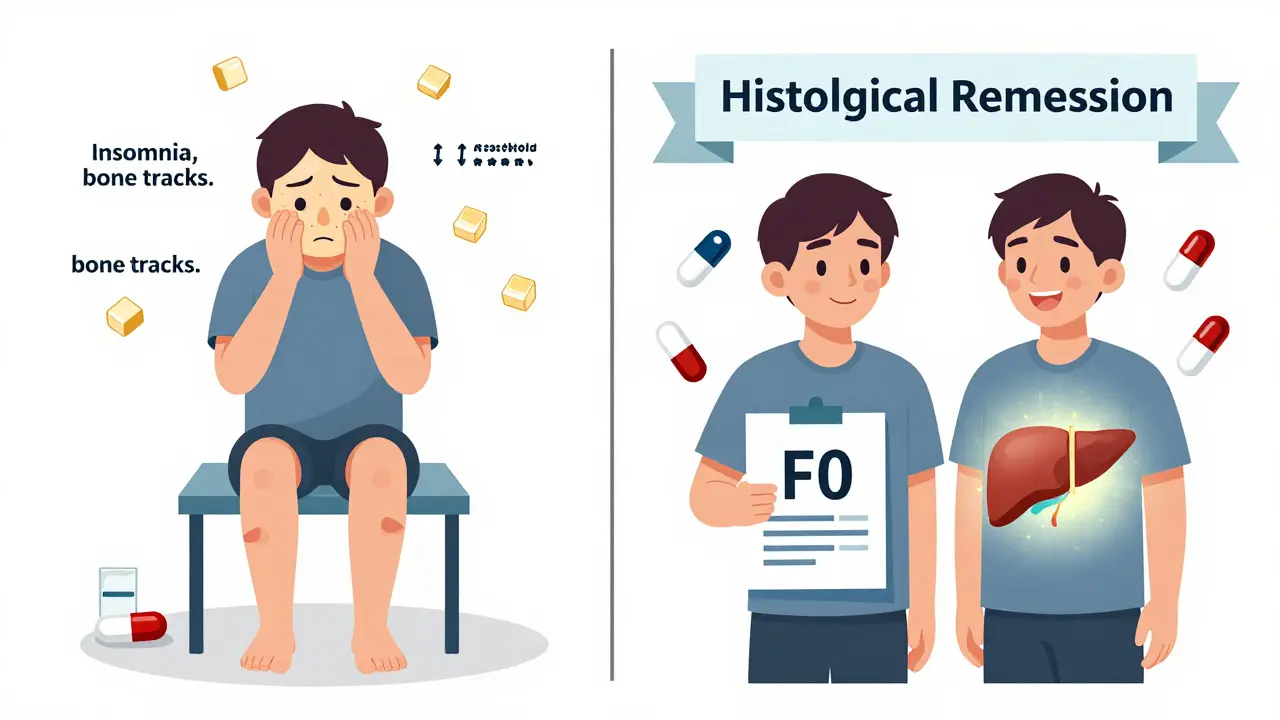
But steroids aren’t harmless. They can cause moon face, weight gain, fluid retention, mood swings, insomnia, and high blood sugar. Within five years, 30-40% of people on long-term steroids develop complications like diabetes (15%), osteoporosis (20%), or cataracts (10%). That’s why doctors don’t keep patients on high doses forever.
The standard starting dose is 0.5 to 1 mg per kilogram of body weight per day-usually around 40-60 mg daily. After four to eight weeks, the dose is slowly lowered. The goal is to get down to 10-15 mg per day by week eight. That’s not easy. Tapering too fast can cause a flare-up. Tapering too slow means more side effects. Most patients need careful monitoring every two to four weeks during this phase, with liver enzyme and IgG levels checked to track progress.
The Role of Azathioprine: Steroid-Sparing and Long-Term Control
Azathioprine (sold as Imuran or generic azathioprine) isn’t used alone-it’s paired with steroids from the start. Its job is simple: let you take less steroid. By combining the two, doctors can reduce prednisone doses by 70-80% within six months. That cuts steroid side effects nearly in half. While 70% of people on steroids alone develop serious side effects, that number drops to 30% when azathioprine is added.
Azathioprine works by suppressing immune cell activity. It’s taken daily, usually starting at 50 mg and increasing to 1-2 mg per kg of body weight (up to 150 mg). It doesn’t work fast. It takes months to show its full effect. But once it does, it helps keep the disease quiet for years.
But azathioprine has risks too. About 35% of patients get nausea, vomiting, or abdominal pain. A small group-around 12%-experience bone marrow suppression, which can lower white blood cells and increase infection risk. That’s why testing for thiopurine S-methyltransferase (TPMT) enzyme levels is now standard before starting. People with a genetic deficiency in TPMT can’t break down azathioprine properly and face a 30-fold higher risk of life-threatening bone marrow failure. Testing costs $250-$400 in the U.S., and while 89% of U.S. academic centers now do it routinely, only 45% of clinics outside academic settings still skip it. Don’t let that happen to you.

What Happens After Treatment Starts?
Complete biochemical response-meaning ALT, AST, and IgG levels return to normal-takes 18 to 24 months. About 60-80% of patients get there. The rest have an incomplete response, with liver enzymes still elevated above twice the normal limit. For them, doctors may switch to second-line drugs like mycophenolate mofetil (CellCept) or calcineurin inhibitors. Mycophenolate works well for people who can’t tolerate azathioprine. In fact, some patients, like one Reddit user who developed pancreatitis on azathioprine, found relief only after switching.
Healing isn’t just about blood tests. The real win is histological remission-meaning the liver tissue itself stops being damaged. A repeat biopsy after two to three years of treatment often shows the inflammation has cleared, and even fibrosis (scarring) can improve. One patient reported their liver fibrosis stage dropped from F3 (moderate scarring) to F0 (no scarring) after two years on low-dose steroids and azathioprine. That’s not rare. Studies show 50-70% of patients achieve this level of healing.
But here’s the catch: most people need to stay on medication long-term. About 60-80% of patients require maintenance therapy. If you stop treatment, there’s a 50-90% chance the disease comes back. Even if you’ve been in remission for two or three years, stopping too soon can trigger a flare within three months. That’s why doctors urge patience and stick to the plan.
What You Need to Know Before Starting Treatment
Before you begin steroids or azathioprine, your doctor must check for hepatitis B. About 15-20% of people carry the virus silently (occult infection). When you suppress your immune system with these drugs, the virus can reactivate-sometimes with deadly results. Testing for hepatitis B surface antigen (HBsAg) and core antibody (anti-HBc) is mandatory. If you’re positive, you’ll start antiviral therapy like tenofovir before starting immunosuppressants.
You should also get vaccinated for hepatitis A and B if you haven’t already. Once you’re on immunosuppressants, vaccines don’t work as well. In healthy people, the hepatitis B vaccine is 90% effective. In those on azathioprine or steroids, it drops to 40-60%. Getting vaccinated before treatment starts gives you the best shot at protection.
Monitoring is ongoing. Blood tests every 2-4 weeks in the first few months, then every three months once stable. IgG levels are checked quarterly. Liver biopsies are recommended after 18-24 months to confirm healing. And if you’re on azathioprine, your blood count needs regular checks for signs of low white cells or platelets.
What’s New in 2025? Updates That Matter
The 2025 EASL guidelines brought major changes. First, they dropped the old system of classifying AIH by autoantibody type. No more splitting patients into Type 1 and Type 2 for treatment purposes. Second, they extended the window for assessing treatment response from six months to six to twelve months. That’s important because some people take longer to respond. Rushing to change treatment too early can lead to unnecessary switches.
Second-line drugs are getting more attention. Mycophenolate mofetil is now a standard alternative to azathioprine, especially for those who can’t tolerate it. And new drugs are on the horizon. JAK inhibitors like tofacitinib and monoclonal antibodies targeting interleukin-6 are showing promise in early trials. One phase 2 trial found clazakizumab led to a 50% complete response rate in 24 weeks. While these aren’t approved yet, they’re giving hope to people who don’t respond to current options.
Even more exciting? Research into biomarkers. Scientists are testing blood panels of microRNAs that might predict who will respond to steroids within just two weeks. If validated, this could save people months of trial and error. Genetic markers like HLA-DRB1*03:01 and *04:01 are already linked to more severe disease, and future treatments may be tailored based on your DNA.
Real-Life Challenges: The Human Side of Treatment
Behind the numbers are real people. One patient on Reddit described steroid side effects as making her ‘unrecognizable’-moon face, 30 pounds of fluid weight in three weeks, and insomnia that felt permanent. Another said azathioprine gave her pancreatitis. These aren’t rare stories. The 2024 Global Autoimmune Institute registry of 1,247 patients found 68% experienced at least one treatment-related side effect. Weight gain, mood changes, and hair thinning were common with steroids. With azathioprine, it was fatigue, nausea, and bone marrow suppression.
But there’s hope. Many patients, once stabilized, report feeling better than they have in years. One person wrote: ‘After two years on 5mg prednisone and 75mg azathioprine, my biopsy showed complete fibrosis reversal.’ That’s not just a lab result-it’s a life regained.
Adherence is a big issue. Only 55% of patients stick with monotherapy (steroids alone), but 75% stay on combination therapy. Why? Because they feel better and have fewer side effects. Support groups and patient education matter. The American Liver Foundation and Autoimmune Hepatitis Association are seeing growing membership-up 20% annually since 2020. More people are learning they’re not alone.
What Comes Next? The Road Ahead
Autoimmune hepatitis is no longer a death sentence. With early diagnosis and consistent treatment, most people live normal lifespans. The goal isn’t just to control symptoms-it’s to heal the liver. And for many, that’s possible.
But it takes patience. It takes trust in your medical team. It takes sticking with the plan even when the side effects are tough. And it takes knowing that what works for one person might not work for another. Your treatment is personal. Your response is unique. Your liver can heal-if you give it the right tools and time.
Can autoimmune hepatitis be cured?
Autoimmune hepatitis isn’t typically cured, but it can be effectively controlled. Most people achieve remission with medication and can live normal lives. About 60-80% of patients reach biochemical remission within two years. However, stopping treatment often leads to relapse-50-90% of patients flare up after discontinuation. Long-term maintenance therapy is needed for 60-80% of people to prevent disease return.
Why do I need a liver biopsy if my blood tests are high?
Blood tests show liver damage, but they don’t tell you why. Many conditions-viral hepatitis, fatty liver, drug reactions-can raise liver enzymes. A biopsy looks at the actual tissue to find the telltale sign of autoimmune hepatitis: interface hepatitis. This specific pattern of inflammation at the liver’s edge confirms the diagnosis. Without it, you risk being misdiagnosed or undertreated.
Can I stop taking steroids and azathioprine after my liver improves?
Some patients try to stop after 2-3 years of remission, but it’s risky. Only about 45% succeed without relapse. Most relapses happen within three months of stopping. If you and your doctor decide to attempt withdrawal, it must be done slowly-over 6-12 months-with close monitoring. Blood tests and sometimes repeat biopsies are needed to catch early signs of return.
Is azathioprine safe if I have a history of cancer?
Azathioprine suppresses the immune system, which can increase cancer risk over time-especially skin cancer and lymphoma. If you’ve had cancer in the past, your doctor will weigh the benefits against the risks. In many cases, they’ll still use it but with extra monitoring, like annual skin checks. Alternatives like mycophenolate may be preferred if the risk is too high.
How do I know if my treatment is working?
Your doctor will track your ALT and AST levels every few weeks at first, then every three months. Normal levels mean the inflammation is under control. IgG levels also drop as the immune system calms. But the best sign of true healing is a repeat liver biopsy after 18-24 months. If the tissue shows no interface hepatitis, you’re in histological remission-this is the gold standard for treatment success.
What if I can’t tolerate azathioprine?
Mycophenolate mofetil (CellCept) is the most common alternative. It’s taken twice daily and works well for people with GI side effects or bone marrow suppression from azathioprine. Other options include calcineurin inhibitors like tacrolimus or cyclosporine. In refractory cases, newer drugs like JAK inhibitors are being tested in clinical trials. Your doctor will choose based on your symptoms, lab results, and genetic testing.
Do I need to avoid alcohol completely?
Yes. Even small amounts of alcohol can accelerate liver damage in autoimmune hepatitis. The liver is already under attack from your immune system-adding alcohol puts extra stress on it. Complete abstinence is strongly recommended. This isn’t just about preventing cirrhosis; it’s about giving your liver the best chance to heal.
Are there any new treatments on the horizon?
Yes. Clinical trials are testing drugs like tofacitinib (a JAK inhibitor) and clazakizumab (an IL-6 blocker), both showing strong results in early studies. Researchers are also developing blood tests that predict steroid response within two weeks using microRNA markers. These could personalize treatment faster and reduce trial-and-error. While not yet approved, they represent the next step in managing autoimmune hepatitis more precisely.




William Minks
8 March, 2026 . 00:18 AM
Just wanted to say this post saved my life. I was misdiagnosed for 2 years with NAFLD until I found this. Started steroids + azathioprine 8 months ago and my ALT dropped from 480 to 32. Still have moon face but hey, my liver is healing 😊
Jeff Mirisola
9 March, 2026 . 19:20 PM
The part about TPMT testing is CRITICAL. My cousin got hit with full bone marrow suppression because they skipped it. Don't be that guy. Get tested. $300 now or ICU later. This isn't optional.
Susan Purney Mark
11 March, 2026 . 01:21 AM
I'm so glad you mentioned the biopsy. I was terrified of the procedure but it was way less bad than I thought. The tech was super calm and gave me a blanket. Two years later, my fibrosis went from F3 to F0. It’s not magic-it’s science. 🌱
Andrew Poulin
11 March, 2026 . 04:52 AM
Azathioprine made me puke for 3 weeks. Switched to mycophenolate. No nausea. Same results. If you're struggling with side effects don't suffer in silence. Talk to your doc. There are options
Sean Callahan
12 March, 2026 . 10:16 AM
i just wanna say i got diagnosed last year and i was so scared. then i read this and i felt like someone finally got it. i cry every time i see my labs are normal now. thanks for writing this. you're a real one 💛
Ferdinand Aton
13 March, 2026 . 14:05 PM
You say steroids are the gold standard but have you considered that maybe the immune system isn't the problem? Maybe it's gut dysbiosis? Or environmental toxins? Just saying…
Ian Kiplagat
14 March, 2026 . 14:59 PM
TPMT testing. Non-negotiable. Saw a case in London where they skipped it. Patient lost 80% of marrow. Ended up in transplant. This isn't theoretical.
Amina Aminkhuslen
15 March, 2026 . 22:39 PM
People who skip the biopsy are gambling with their liver. It’s not a suggestion-it’s a lifeline. If your doc won’t order it, find a new one. This isn’t the 90s anymore.
amber carrillo
17 March, 2026 . 09:06 AM
I appreciate the clarity and evidence-based approach. It's rare to find a medical post this thorough without sounding clinical. Thank you for sharing.
Tim Hnatko
17 March, 2026 . 20:45 PM
I've been on 5mg prednisone and 75mg azathioprine for 3 years. My biopsy last month showed zero inflammation. I'm not cured but I'm alive. And that's enough.
Aaron Pace
19 March, 2026 . 20:30 PM
I got the biopsy and it was fine. Then I found out I have a rare TPMT variant. They switched me to mycophenolate. I'm now 18 months in and my numbers are perfect. You're not alone. 🙌
Joey Pearson
19 March, 2026 . 22:13 PM
You got this. One day at a time. Labs don't define you. Your courage does. Keep going. You're stronger than you think 💪