Imagine you've been taking a specific medication for years with no issues. One day, your pharmacy switches you to a cheaper version, and suddenly you feel more drowsy or notice a strange side effect. The first question that pops up is: "Is this generic version reacting differently with my other meds?" It's a common worry, especially when you're managing multiple prescriptions. The short answer is that for the vast majority of people, the risk of drug interactions is exactly the same whether you take the brand-name or the generic version.
The Secret to Why Generics Work the Same
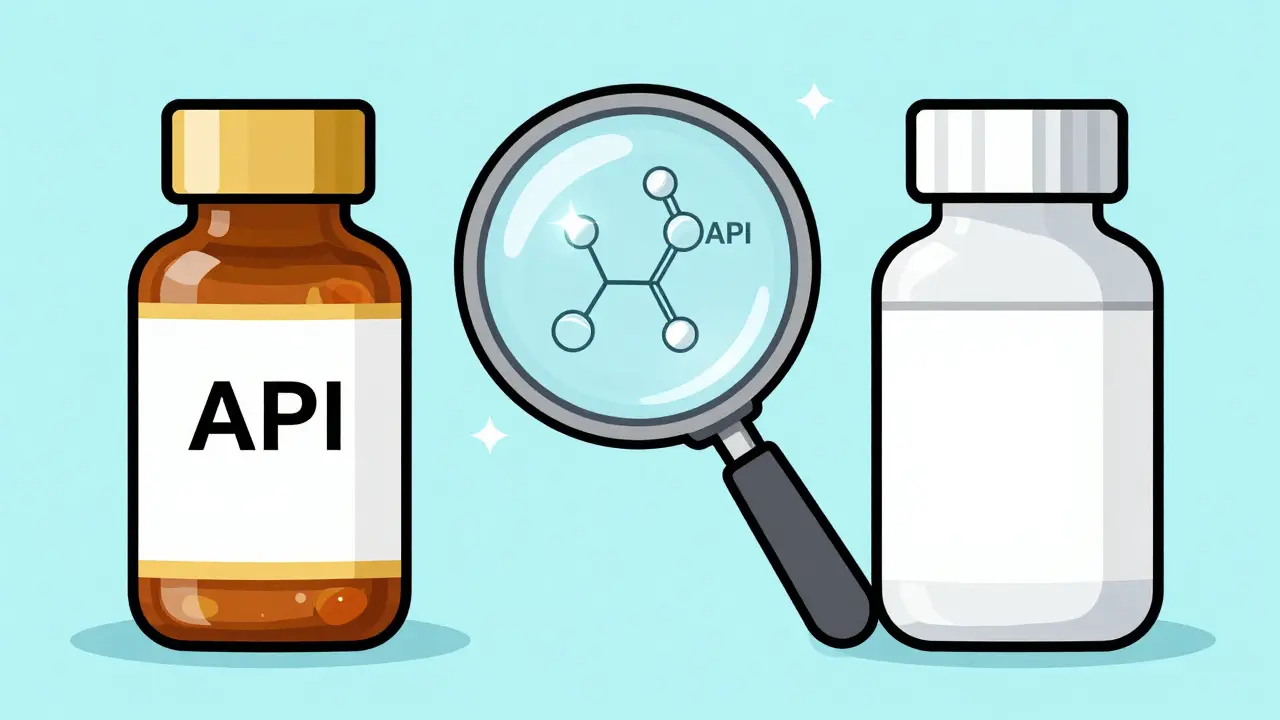
To understand why the risk doesn't change, we have to look at what's actually inside the pill. When the FDA (U.S. Food and Drug Administration) approves a generic drug, they aren't just guessing. They require the manufacturer to prove bioequivalence. This is a fancy way of saying the generic version delivers the same amount of active ingredient into your bloodstream in the same amount of time as the original.
The active pharmaceutical ingredient (API) is the part of the drug that does the heavy lifting-it's what treats your condition and, crucially, it's what causes interactions with other substances. Since the API is identical in both the brand and generic versions, the chemical "conversation" between that drug and another medication in your system remains the same. If a brand-name blood pressure med interacts with a specific diuretic, the generic version will do the exact same thing because the chemistry hasn't changed.
Where Brand and Generic Drugs Actually Differ
If the active ingredient is the same, what's actually different? The answer lies in the excipients. These are the inactive ingredients-things like fillers, binders, coloring agents, and preservatives. While they don't change how the drug treats your illness, they can occasionally cause issues for a very small number of people.
For example, if you have a severe lactose intolerance, a generic pill that uses lactose as a filler might cause stomach upset, whereas the brand-name version might use a different binder. This isn't a "drug interaction" in the chemical sense, but rather a sensitivity to an inactive ingredient. It's rare, but it's the primary reason some people feel a difference when switching brands.
| Feature | Brand-Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Original formulation | Identical to brand |
| Inactive Ingredients (Excipients) | Specific to brand | May vary by manufacturer |
| FDA Bioequivalence Requirement | N/A (The Reference) | Must fall within 80-125% range |
| Primary Interaction Risk | Based on API | Based on API (Same as brand) |
| Cost | Typically higher | Typically significantly lower |

Dealing with Narrow Therapeutic Index (NTI) Drugs
While generics are safe for most, there is a special category called Narrow Therapeutic Index (NTI) drugs. These are medications where a tiny change in the blood concentration can be the difference between the drug working and the drug becoming toxic. Think of things like certain blood thinners or anti-seizure medications.
For these specific drugs, the FDA and other regulators often require tighter bioequivalence ranges. While a standard generic can have a slight variation in absorption, NTI drugs have a much smaller window for error. In these rare cases, your doctor might write "dispense as written" on your prescription. This tells the pharmacist not to switch you to a generic, ensuring you stay on the exact same formulation to avoid any fluctuations in how the drug interacts with your body.

The Psychology of the Switch: Is it a Nocebo Effect?
You'll often find people on forums like Reddit claiming that the generic version "just doesn't work as well" or "gives me more side effects." Interestingly, data suggests that some of this is psychological. There's something called the nocebo effect-the opposite of a placebo. If you believe a generic drug is inferior, your brain can actually manifest physical symptoms or a perceived lack of efficacy.
One study noticed that complaints about generic drugs often spike in the first three months after a new generic enters the market, but then they drop back down. This suggests that once the initial anxiety of switching wears off, people realize the drug is working just fine. When we look at the hard data, such as a study of 17 cardiovascular drugs, generics were found to be at least as effective-and sometimes even associated with lower mortality rates-than brand names.
Practical Tips for Switching Safely
If you're nervous about switching to a generic or if you've noticed a change after a switch, don't panic. Here is a practical approach to handling it:
- Keep a log: Note exactly when you switched and what symptoms you're feeling. This helps your doctor determine if it's a real reaction or a coincidence.
- Check the filler: If you have known allergies or intolerances (like lactose or gluten), ask your pharmacist to check the inactive ingredients in the generic version.
- Stick with one manufacturer: If your pharmacy switches your generic provider every month, you might notice slight differences. You can ask your pharmacist to provide the same manufacturer's version consistently.
- Talk about your "stack": Always make sure your pharmacist has a full list of your supplements, vitamins, and other meds. The interaction risk comes from the combination of drugs, not from whether the drug is generic.
Can a generic drug cause a new interaction that the brand name didn't?
Generally, no. Because the active ingredient is the same, the chemical interactions are identical. The only exception is a very rare reaction to an inactive ingredient (filler), which is an allergy or sensitivity rather than a traditional drug-drug interaction.
Why do some doctors insist on brand-name only?
This usually happens with Narrow Therapeutic Index (NTI) drugs, where the margin for error is tiny. In these cases, switching between different generic manufacturers could cause slight fluctuations in drug levels, which might affect the treatment's stability.
Does the FDA really test generics as much as brands?
Generics don't go through the same massive clinical trials as the original brand-name drug because the safety and efficacy of the active ingredient have already been proven. Instead, they undergo bioequivalence testing to prove they perform the same way in the body.
Are generics in the US the same as generics in Europe?
The goal is the same, but the specific regulations can differ. For instance, the European Medicines Agency (EMA) sometimes requires even tighter bioequivalence ranges for certain high-risk drugs than the FDA does.
What should I do if I feel a difference after switching to a generic?
Contact your pharmacist or doctor. They can check if the generic has a different inactive ingredient that you might be sensitive to, or they can ensure you are using a consistent manufacturer to avoid "formulation jump."




Mark Koepsell
30 April, 2026 . 00:31 AM
The point about bioequivalence is key here. Most people don't realize that the 80-125% range is a statistical window for the rate and extent of absorption, not a random guess. It's a standard pharmacokinetic measure.
Bradley Gusick
1 May, 2026 . 14:20 PM
Sure, just trust the FDA! 🙄 Total joke. They're probably just letting some random overseas factory pump out fillers that we don't even know about. It's all about the money and control. Wake up people, the "bioequivalence" is just a buzzword to keep us compliant while they experiment on us with cheap substitutes!
Joel Bonstell
3 May, 2026 . 00:36 AM
I totally get the stress of switching meds. I've had a few times where a new generic just felt... off. Like, not a full blowen interaction, but just a weird foggy feeling in my head for a week. It's realy helpful to keep a log like this suggests so your doc actually listens to you.
Robert Cowley
4 May, 2026 . 05:12 AM
Oh please, this is just a love letter to Big Pharma's regulatory wing. 🙄 Imagine thinking a "range" of 25% is actually identical. That's a massive variance in any other scientific field! This whole "nocebo" argument is just a convenient way to gaslight patients who actually feel a difference. Pure comedy! 🤡
J. Walter Jenkem
6 May, 2026 . 03:48 AM
It's great that we're discussing this. Focusing on a consistent manufacturer is a really solid piece of advice for anyone feeling unstable during a transition.
Jimmy Crocker
6 May, 2026 . 08:16 AM
One must simply acknowledge that the pharmacological nuance of excipients is often overlooked by the layperson, and while the active moiety remains constant, the pharmacokinetics can be subtly altered by the delivery vehicle, which is why some of us with more refined physiological responses find the generic alternatives to be utterly subpar in their execution, though I suppose for the average person it's adequate enough if one isn't too picky about their health outcomes.
Allison Maier
6 May, 2026 . 11:53 AM
too long didn't read. generics suck 🙄
princess lovearies
7 May, 2026 . 08:43 AM
It's interesting how our minds and bodies dance together in these reactions. Whether it's a physical sensitivity or a mental block, it just shows how connected we are. Be gentle with yourselves if you're feeling a shift.
Srinivas Komakula
8 May, 2026 . 08:45 AM
The systemic failure of the regulatory apparatus is evident!! These bioequivalence parameters are mere facades for the infiltration of sub-standard API synthesis from non-compliant geopolitical zones!! It is an absolute travesty of pharmaceutical oversight!!
Ken Baldridge
10 May, 2026 . 08:39 AM
Let's dive into the pharmacodynamics here. The Cmax and AUC-area under the curve-are the actual metrics we're talking about when we say bioequivalent. If the bioavailability is within that window, the therapeutic index usually stays stable, unless you're dealing with those NTI drugs where the window is tight. Just keep your stack optimized and stay chill!
Preety Singh
11 May, 2026 . 20:54 PM
The assertion that a generic is merely "adequate" is a pedestrian view. True quality is found in the original synthesis. I find it quite quaint that people believe a filler is a negligible detail when it speaks to the very essence of the product's purity.
Leah Sentz
12 May, 2026 . 07:30 AM
I ONLY TRUST AMERICAN MADE MEDS! 🇺🇸💊 Anything else is just garbage from other countries trying to poison us! 😡😤 My pharmacy tried to switch me once and I almost lost my mind! NO THANK YOU! 🇺🇸🇺🇸🇺🇸
nikki paurillo
12 May, 2026 . 21:45 PM
There's a certain poetic irony in how we trust a chemical formula yet fear the vessel it arrives in. It's a tapestry of trust and suspicion woven into our modern healthcare system.
Seema Karanje
12 May, 2026 . 22:08 PM
STOP COMPLAINING AND JUST TAKE THE MEDS! If the doctor says it works, it works! Get over your tiny problems and focus on getting better! NOW!
Lando Neal
14 May, 2026 . 10:39 AM
Wow!! I never knew about the nocebo effect!! That is so wild!! I'm definitely going to try to keep a more positive mindset next time my insurance changes my brand!! Thanks for the info!!